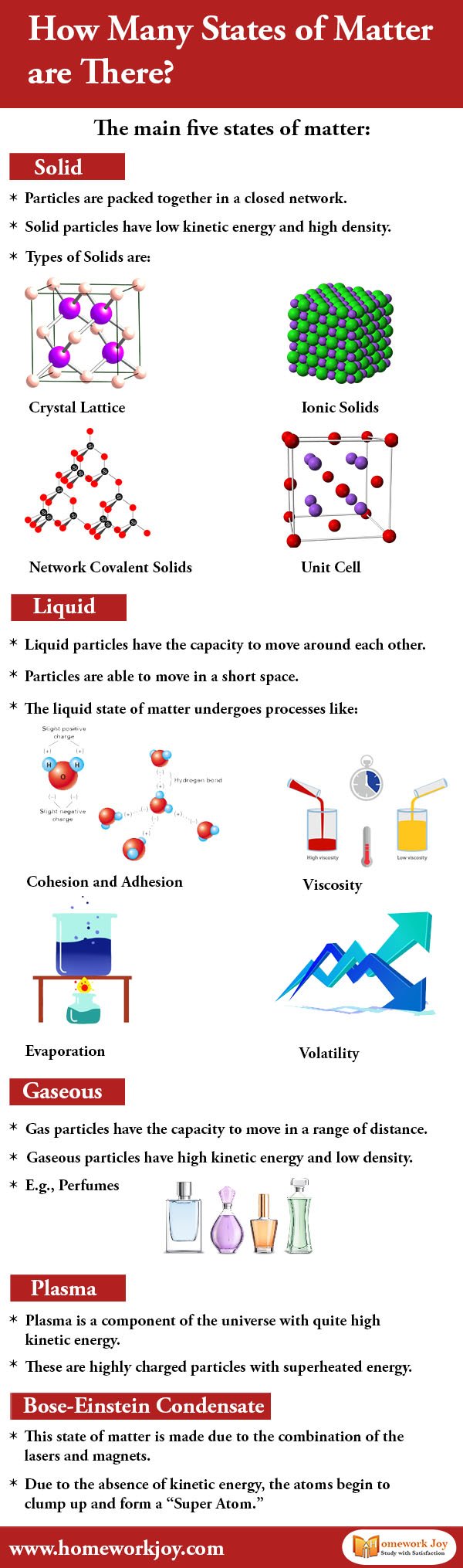
There are five states of matter on the site: Solid, Liquid, Gaseous, Plasma, and Bose-Einstein condensate. All of these are different states of matter that have different physical properties. But do you know how does matter changes from one state to another? Let us explain.
Elements and compounds can move from one state to another when particular physical conditions change. If the temperature of one system increases, then the elements in that system become excited and active. Thus changes their state of matter.
5 States of Matter
Here we will discuss five states of matter one by one.
Solid
Solid particles have low kinetic energy and high density. The forces between these particles are so strong that they can not even move freely but can vibrate. Hence they are packed together in a closed network. They have a fixed shape and definite volume. Solids can only change their shape only by an outside force.
Types of Solids are:
- Crystal Lattice
- Network Covalent Solids
- Ionic Solids
- Unit Cell
Liquid
Liquid particles can move around each other. Particles can move or slide past one another in a short space. Liquids assume the shape of the container in which it occupies. They are less compressible than gaseous molecules. If the temperature and pressure are constant, then the volume is definite.
The liquid state of matter undergoes processes like:
- Cohesion and Adhesion
- Viscosity
- Evaporation
- Volatility
Gaseous
Gas particles can move in a range of distances. They move with no regular arrangement and are highly compressible. The molecules have high kinetic energy and low density. The gas has no definite shape and volume. However, it occupies the whole container in which it is confined.
E.g., Perfumes, nitrogen gas
Plasma
Plasma is a component of the universe with quite high kinetic energy. These are highly charged particles with superheated energy. Just like gas, plasma does not have definite shape and volume. They are electrically conductive. Furthermore, they produce electric currents and magnetic fields.
Bose-Einstein Condensate
In 1924, Albert Einstein and Satendra Nath Bose predicted it. The combination of the lasers and magnets made this state of matter. The matter stops behaving as an independent particle and collapses into a single quantum state. Due to the absence of kinetic energy, the atoms begin to clump up and form a “Super Atom.”